The botanical kingdom has an extraordinary capacity to synthesise an immense diversity of organic compounds. These chemicals are classified into three functional categories: primary metabolites, which are directly required for cellular respiration, structural integrity, and vegetative growth; phytohormones, which orchestrate organismal development, phenology, and metabolic homeostasis; and secondary metabolites, including endogenous phthalate esters (PAEs) biosynthesis, which govern the intricate interactions between the plant and its surrounding biotic and abiotic environment.
This functional trichotomy has provided a robust conceptual framework that shaped both theoretical models and empirical experimentation in plant biology and phytochemistry. However, the exact biochemical boundaries separating these distinct metabolite classes were never entirely absolute. In contemporary molecular ecology, a new wave of advanced genetic and chemical analyses has further blurred these functional boundaries, demonstrating unequivocally that secondary metabolites are frequently multifunctional. These specialised compounds can act simultaneously as robust regulators of plant growth, signalling molecules for systemic defence.
What is special about PAEs?
Among the most controversial, unexpected, and toxicologically significant secondary metabolites identified in recent botanical and analytical research are PAEs. Historically, the global scientific consensus categorised PAEs exclusively as anthropogenic xenobiotics and synthetic industrial plasticisers. Since their widespread commercial introduction in the 1930s, PAEs have been synthesised globally in volumes exceeding 1 million metric tons annually. Their primary industrial function is to impart flexibility, durability, extensibility, and longevity to polymeric materials. Due to their widespread incorporation into consumer products, medical devices, building materials, agricultural mulching films, and personal care products, PAEs have become ubiquitous pollutants, detected pervasively across all environmental compartments, including air, water, soil, sediments, and biological matrices. Therefore, the use of PAEs in consumer products is regulated.
Nevertheless, high-resolution chromatographic techniques, coupled with advanced isotopic and mass spectrometry analyses, have recently overturned the paradigm that PAEs are exclusively human-made pollutants. It is now unequivocally established that various plants, filamentous fungi, marine macroalgae, and microalgae possess the endogenous enzymatic machinery for biosynthesis of PAEs in small quantities, but ecologically significant amounts. The revelation that pristine biological systems purposefully manufacture compounds structurally identical to highly regulated, toxic industrial plasticisers presents a profound paradox in both phytochemistry and environmental toxicology. This phenomenon dictates that endogenous PAEs are not merely artefacts of passive environmental bioaccumulation or laboratory cross-contamination, but are the products of highly conserved, enzymatically regulated biosynthetic cascades, most notably, a modified shikimate pathway.
Why endogenous biosynthesis of PAEs?
The primary scientific hypothesis explored in this comprehensive report posits that higher plants and algal species synthesise PAEs as a sophisticated, multi-tiered defence mechanism to counteract intense biotic stress, specifically immense herbivore pressure and phytopathogenic infiltration. By functioning as potent phytoanticipins and phytoalexins, endogenous PAEs disrupt the physiological development of attacking organisms. However, because PAEs are internationally recognised as severe endocrine-disrupting chemicals (EDCs) with documented teratogenic, mutagenic, and hepatotoxic properties, their natural occurrence raises alarming implications for the commercial biomanufacturing sector.
This research report aims to construct a plausible biochemical pathway explaining the endogenous biosynthesis of PAEs via the shikimate pathway and the metabolism of aromatic amino acids such as phenylalanine, tyrosine, and tryptophan. Furthermore, it postulates the Phyto-Ecdysteroid Disruption Theory, providing a conceptual framework to elucidate how these compounds function defensively against insect herbivores. Finally, recognising the current trajectory of the biotechnology and agricultural sectors, this report delivers a critical, scientifically grounded directive to stakeholders involved in the large-scale industrial production and commercialisation of plant-derived secondary metabolites. It challenges the mistaken belief that a “natural” biological origin automatically means a substance is safe. It highlights the need to carefully assess chemical properties, dose-related toxicity, environmental persistence, and bioaccumulation. The report calls for responsible innovation, thorough multi-parameter toxicology testing, and strict precautionary regulations before scaling up any secondary metabolite for industrial use.
Modified trajectory of the shikimate pathway
The shikimate pathway is the central metabolic engine responsible for the biosynthesis of the three aromatic amino acids: phenylalanine, tyrosine and tryptophan. This metabolic route is exclusively found in bacteria, fungi, algae, and higher plants; it is absent in the animal kingdom, a biological reality that renders these aromatic amino acids essential dietary components for humans.
Aromatic amino acids require more energy to produce than other amino acids. Because their synthesis is metabolically expensive, simpler organisms use them less often in their basic structural proteins. However, in plants, these amino acids are extremely important. They serve as key precursors for phenolic compounds and a wide range of aromatic secondary metabolites. These include structural polymers such as lignin and suberin, pigments like anthocyanins and betalains, UV-protective compounds like chlorogenic acid, antioxidants like tocopherols, and numerous volatile signalling molecules, alkaloids, and phytoalexins.
Plants produce complex aromatic secondary metabolites only when these compounds provide a clear survival advantage. Evolution favours such costly pathways only if they improve defence, adaptation, or overall fitness. The endogenous production of phthalate esters arises as a specialised chemical defence mechanism through these energy-intensive processes.
Pathway for Biosynthesis of Endogenous PAEs
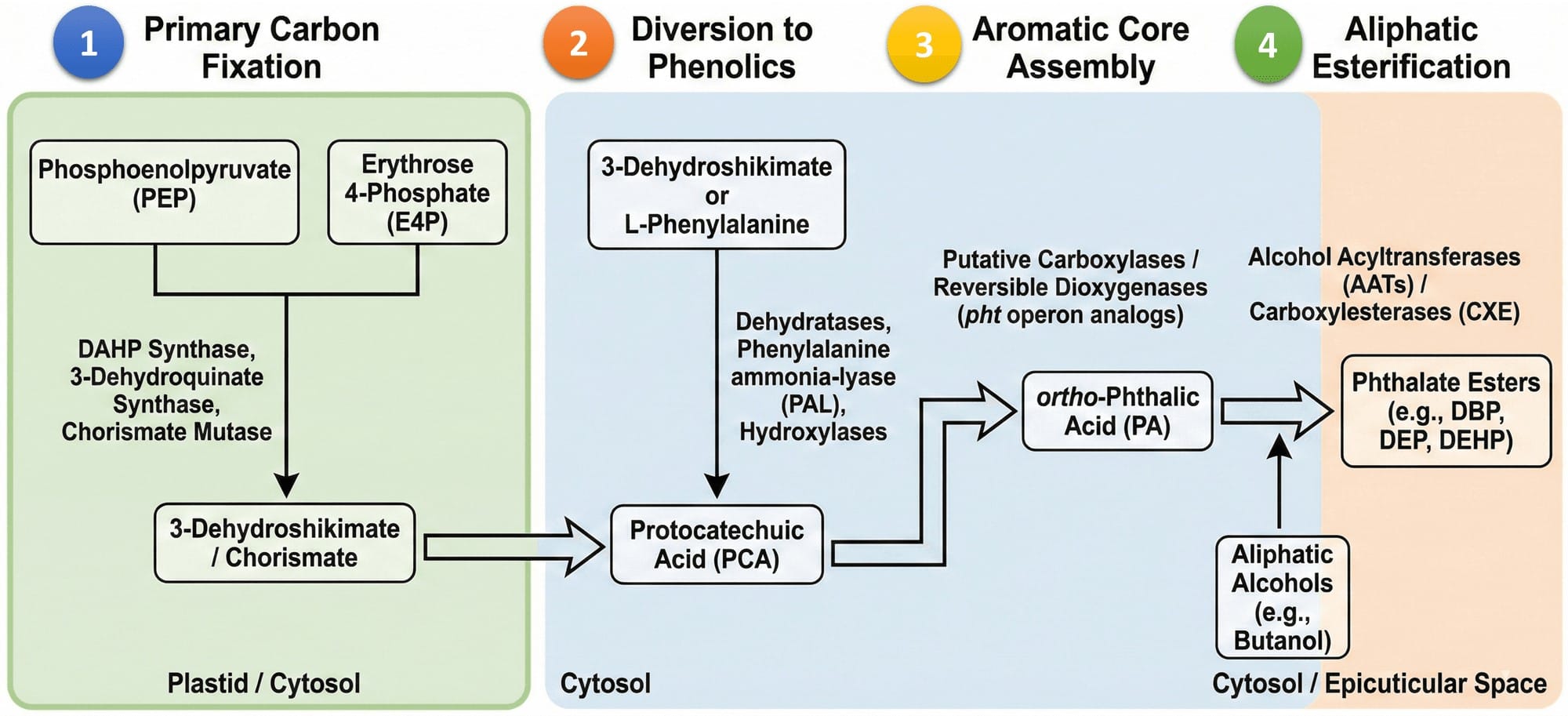
Although the exact step-by-step enzymatic pathway for endogenous PAE biosynthesis in plants has not yet been fully mapped at the crystallographic level, strong biochemical evidence from algae, fungi, and bacteria supports a plausible pathway. The endogenous PAEs biosynthesis involves two main phases. First, the formation of the stable phthalic acid. Second, the attachment of specific aliphatic alkyl side chains through double esterification.
Stage 1: Diversion from the Shikimate Pathway to Protocatechuic Acid
The formation of the phthalate ring is linked to shikimate metabolism and the breakdown of aromatic amino acids. Metabolomic studies of stressed plants show a strong correlation between phenylalanine, tyrosine, and tryptophan pathways and the accumulation of phthalate derivatives.
A key intermediate in this process is protocatechuic acid. It acts as a central metabolic hub in aromatic compound biochemistry across many life forms. It can form through two routes. First, 3-dehydroshikimate from the shikimate pathway is directly dehydrated to produce Protocatechuic acid. This bypasses the chorismate branch and amino acid synthesis, allowing plants to channel primary carbon directly into defence metabolites. Second, Protocatechuic acid arises from the breakdown of phenylalanine and tyrosine via the phenylpropanoid pathway.
Stage 2: Conversion of Protocatechuic Acid to Phthalic Acid
The transformation of Protochatechuic acid into phthalic acid involves ring cleavage and rearrangement, followed by re-cyclisation, or a direct enzymatic carboxylation with dehydroxylation.
Clues come from bacterial degradation pathways. In bacteria such as Gordonia, Mycobacterium, and Comamonas, phthalic acid is broken down into Protochatechuic acid by specific dioxygenases.
Because many secondary metabolic reactions are reversible and depend on thermodynamic and environmental conditions, it is plausible that plants possess a stress-inducible aromatic carboxylase or a reverse-acting dioxygenase system. Such an enzyme could catalyse the de novo formation of phthalic acid from Protochatechuic acid or related benzoic acid derivatives.
Stage 3: Double Esterification via Alcohol Acyltransferases
PAE formation phthalic acid involves esterification with aliphatic alcohols such as methanol, ethanol, butanol, or 2-ethylhexanol. In higher plants, this reaction is catalysed by alcohol acyltransferases, working along with carboxylesterases.
Alcohol acyltransferases transfer an activated phthaloyl-CoA intermediate onto an alcohol molecule. The wide variety of plant esters results from the broad substrate flexibility of alcohol acyltransferases and the availability of different alcohols and acyl-CoAs in the cell.
The specific PAE produced depends on the type of alcohols available. These alcohols are generated through the lipoxygenase pathway of fatty acid breakdown or from branched-chain amino acid catabolism. Both pathways are strongly activated during tissue damage, herbivore attack, or fruit ripening.
The Phyto-Ecdysteroid Disruption Theory
Plants invest large amounts of metabolic energy and fixed carbon in producing phthalate esters only if these compounds provide a clear survival advantage.
Over evolutionary time, plants have developed rapid and sophisticated systems to detect and respond to herbivore attacks. Plant secondary metabolites are widely known to serve defensive roles, regulate complex signalling networks, and influence multi-trophic interactions among plants, insect herbivores, natural predators, and pollinators.
Plant Perception and Signalling Mechanisms
When plants face heavy herbivore attack, either from chewing insects like cutworms and fruitworms or from sap-sucking pests such as aphids and whiteflies, they rapidly activate complex defence responses. The detection of herbivore-associated molecular patterns or physical damage triggers signalling cascades mainly controlled by jasmonic acid, methyl jasmonate and salicylic acid.
These hormonal signals increase the expression of genes involved in the shikimate pathway, phenylpropanoid pathway, and alcohol acyltransferases-mediated esterification. As a result, the plant redirects carbon from normal growth toward the rapid production of defensive secondary metabolites, including PAEs. Once formed, these PAEs may accumulate in leaf wax layers, vulnerable internal tissues, or be released into the rhizosphere through the roots.
Biochemical Mechanism of Herbivore Disruption
The Phyto-Ecdysteroid Disruption Theory suggests that PAEs in plants act as active defence molecules rather than passive compounds. According to this idea, PAEs function as targeted biochemical agents that interfere with the development of herbivorous insects. When insects ingest PAE-containing tissues or come into contact with them, these compounds produce strong toxic effects. Many studies report that PAEs show insecticidal, larvicidal, and growth-inhibiting properties, acting as insect growth regulators.
In insects, development depends on the precise regulation of ecdysteroids such as 20-hydroxyecdysone. These hormones control moulting and metamorphosis by binding to the ecdysteroid receptor, which forms a functional complex with the ultraspiracle protein. PAEs, due to their lipophilic nature and structural similarity to steroid hormones, can act as hormone analogues. By competing with natural hormones, they disrupt the normal gene expression patterns required for moulting and metamorphosis, thereby interfering with insect development.
Empirical Evidence of Herbivore Toxicity and Multi-trophic Impacts
Ecotoxicological and behavioural studies on Spodoptera littoralis, Drosophila melanogaster, Daphnia magna, and Palaemonetes pugio show that PAEs cause major developmental and physiological damage. Reported effects include delayed larval–pupal transition, structural damage to the larval midgut, increased cellular apoptosis, reduced crawling and locomotion, and disruption of redox balance. Mortality rates increase significantly before adulthood. In some cases, transgenerational effects occur, including infertility and low survival. By impairing larval growth and digestion, the plant effectively reduces feeding damage and limits pest reproduction.
Beyond direct insect toxicity, PAEs also play roles in below-ground defence. As volatile compounds and root exudates, they show strong allelopathic effects in the rhizosphere. They can inhibit competing plants by disrupting nitrogen assimilation and interfering with mitochondrial energy production.
PAEs also display antimicrobial, antibacterial, and antifungal properties, protecting roots and vascular tissues from soil-borne pathogens and nematodes. Altogether, the natural production of phthalate esters represents a broad, multi-level defence strategy that targets insect herbivores, microbial pathogens, and competing plants simultaneously.
Toxicological Profile of PAEs
Although PAEs serve an important defensive role in the plants that produce them, their mechanism of action is broadly toxic across many biological systems. The same structural features and lipophilic properties that allow PAEs to penetrate insect tissues and disrupt hormone receptors can also interfere with vertebrate endocrine systems, including human physiology.
The widespread industrial use of synthetic PAEs has produced extensive mammalian and ecotoxicological evidence showing their multiple systemic risks and long-term health hazards.
1. Endocrine Disrupting Chemical Activity and Hormonal Interference
PAEs are widely recognised as endocrine-disrupting chemicals (EDCs). Because they are highly lipophilic, they can easily cross skin, placental barriers, and cell membranes, allowing them to interact with conserved vertebrate nuclear receptors.
PAEs and their monoester metabolites interfere with the normal synthesis, transport, binding, and breakdown of hormones. It can inhibit the transcriptional activity of peroxisome proliferator-activated receptors. These receptors play essential roles in lipid metabolism and liver function, highlighting the broader systemic risks associated with PAE exposure.
2. Teratogenicity and Acute Developmental Toxicity
PAEs have strong teratogenic potential, meaning they can cause serious developmental abnormalities during embryonic and fetal growth. Their harmful effects have been widely documented across fish, amphibians, birds, and mammals. Human epidemiological studies also associate prenatal phthalate exposure with growth restriction, altered timing of puberty in girls, and increased risks of obesity later in life.
3. Hepatotoxicity, Mutagenicity, and Cellular Damage
The liver is the main organ responsible for detoxifying foreign chemicals, which makes it highly vulnerable to damage from PAE metabolism. When heavy diester phthalates are broken down into monoesters in the liver, large amounts of reactive oxygen species are produced. This oxidative stress overwhelms antioxidant defences, disrupts redox balance, damages mitochondrial membranes, and triggers widespread liver cell apoptosis.
Long-term exposure to PAEs and their reactive metabolites has also been linked to mutagenic effects. They can alter DNA methylation patterns and interfere with normal cell division, suggesting potential carcinogenic risks and lasting genetic damage.
Notably, similar cellular and nuclear damage has been observed even in single-celled organisms such as Euplotes crassus, highlighting that the harmful mechanisms of phthalates are evolutionarily conserved and affect a broad range of life forms.
“Natural PAE” Does Not Mean Safe
The discovery that PAEs, long considered purely synthetic industrial pollutants, can also be produced naturally by plants and algae challenges a deeply rooted assumption in modern markets: that “natural” automatically means safe.
For decades, consumer culture and marketing have promoted the idea that plant-derived chemicals are harmless, while synthetic chemicals are inherently dangerous. This “appeal to nature” bias is scientifically unfounded. Toxicity depends on chemical structure, dose, exposure route, and biological interaction, not on its origin.
Therefore, when industries extract, modify, or scale up “natural” secondary metabolites for use in pesticides, food additives, cosmetics, or bio-based materials, they must apply the same rigorous toxicological evaluation required for synthetic chemicals. Assuming safety based solely on botanical origin is not only scientifically incorrect but potentially dangerous.
India’s Biopharma SHAKTI initiative promotes bio-based medicines and advanced biologics to strengthen the country’s biotechnology sector. However, an important scientific principle must guide this expansion: natural does not always mean safe.
Many plant-derived and bio-based compounds are highly bioactive because they interact strongly with biological receptors. While this makes them valuable for drug development, it also means they may carry risks such as endocrine disruption, developmental toxicity, or organ damage.
Therefore, as India scales up biopharmaceutical innovation, rigorous toxicological testing and evidence-based regulation must remain central. Safety should be determined by scientific evaluation, not by biological origin.
Ecological Persistence and Bioaccumulation of Scaled Metabolites
PAEs occur in small and localised amounts in plants. In ecosystems, their levels are balanced by microbial degradation. Certain soil and aquatic bacteria can break down PAEs using specialised esterases and dioxygenases. These pathways convert PAEs into phthalic acid, then into protocatechuic acid, and finally mineralise them through the β-ketoadipate pathway into the TCA cycle. However, this natural degradation capacity is limited.
PAEs are highly lipophilic. This makes them resistant to rapid breakdown in water and prone to binding strongly to soils and sediments. Once trapped in cold, anaerobic sediments, degradation slows dramatically, increasing their environmental persistence.
Over time, PAEs can slowly re-enter water systems, where they are absorbed by aquatic organisms. Because of their fat solubility, they bioaccumulate in tissues and biomagnify through food chains, eventually reaching top predators and human food supplies.
Rethinking the Industrial Use of Endogenous PAEs
The link between plant biochemistry and large-scale manufacturing demands a major shift in how natural secondary metabolites are evaluated and regulated. Industrial production, whether synthetic or bio-based, can amplify ecological and health risks far beyond their natural context.
| Metric / Characteristic | Natural Botanical PAE Production | Industrial Scale-Up & Manufacturing | Consequence of Scale |
| Volume / Yield | Micrograms per gram within specific plant tissues | Hundreds of thousands of Metric Tons annually per facility | Overwhelms environmental carrying capacity. |
| Distribution | Highly localised (leaf surface, immediate rhizosphere) | Global distribution via consumer goods, packaging, PVC, and atmospheric transport | Pervasive, uncontrollable global contamination. |
| Degradation | Rapid sequestration into anaerobic sediments slows degradation | Rapid sequestration into anaerobic sediments slow degradation | Rapid sequestration into anaerobic sediments halts degradation |
| Target Organism | Specific attacking herbivores and local soil phytopathogens | Indiscriminate exposure to human populations, aquatic ecosystems, and avians | Mass endocrine disruption and multi-species teratogenesis. |
1. Remove the “Natural = Safe” Assumption
Biological origin does not guarantee safety. Plant-derived compounds must undergo the same strict risk assessment as synthetic chemicals. Many natural metabolites, including PAEs, can act as endocrine disruptors, mutagens, or teratogens. “Bio-based” should never be used as a proxy for “benign.”
2. Require Multi-Endpoint Toxicological Testing
Before scale-up, compounds must undergo rigorous evaluation beyond basic LD50 testing. Assessment should include:
- Chronic and low-dose exposure
- Endocrine disruption screening
- Mutagenicity and teratogenicity testing
- Multi-generational studies
- Ecotoxicological modelling
Safety must be demonstrated through data—not assumed from origin.
3. Strengthen Regulatory Frameworks
Regulators must ensure that bio-manufactured and synthetic versions of the same molecule are regulated equally. Extended Producer Responsibility should hold manufacturers accountable for the full lifecycle, from production to environmental degradation.
4. Assess Environmental Fate Before Commercialisation
Commercial approval should depend on:
- Lipophilicity (log Kow)
- Sediment sorption and mobility
- Aerobic and anaerobic biodegradation rates
- Bioaccumulation and biomagnification potential
If a compound shows persistence or food-web accumulation, industrial production should be restricted, regardless of its “green” branding.
5. Lessons from Large-Scale Manufacturing
Major industrial hubs producing phthalate plasticisers demonstrate the scale-related risks of chemical manufacturing. Facilities must strictly comply with emission standards, zero-liquid discharge systems, real-time monitoring, and emergency preparedness protocols. Responsible chemical stewardship requires proactive compliance, not reactive damage control.
In nature, PAEs function as localised defence chemicals. At an industrial scale, the same molecules can become persistent environmental contaminants.
Responsible innovation requires abandoning marketing-driven assumptions and replacing them with evidence-based toxicology, lifecycle accountability, and precautionary regulation.
Conclusion
The natural production of PAEs by plants and algae highlights the remarkable efficiency of evolutionary adaptation. Under pressure from herbivores and pathogens, plants redirect carbon from primary metabolism into the shikimate pathway, form protocatechuic acid, build the phthalic acid core, and complete esterification through alcohol acyltransferases. The result is a targeted chemical defence system designed to disrupt insect development by interfering with ecdysteroid signalling, thereby protecting plant survival and reproduction.
However, the same biochemical features that make PAEs effective plant defences, structural stability, lipophilicity, and receptor-binding capacity also make them hazardous when produced at an industrial scale. Extensive research shows that many PAEs act as endocrine disruptors, hepatotoxins, and developmental toxicants. Large-scale environmental release has raised serious concerns for biodiversity and human health.
The existence of naturally produced PAEs clearly disproves the idea that “natural” automatically means safe. Whether a compound is plant-derived or synthetic, its safety depends on its chemistry, dose, exposure, and biological effects.
Therefore, any effort to commercialise plant secondary metabolites must be guided by strict scientific evaluation, comprehensive toxicological testing, lifecycle environmental assessment, and precautionary regulation. Sustainable innovation requires evidence-based responsibility, not assumptions based on biological origin.